Manganese Ore and Make-Up

The ore of manganese is silver-gray with a pink like tinge. It is a tough magnetic element, but can be very brittle. Pyrolusite, this mineral is the main one used in manganese ore. Manganese ore is both produced and found in South Africa and Ukraine. These two places hold up to 80% of the manganese ore reserves.
Complete Electron Configuration Notation for the Atom:

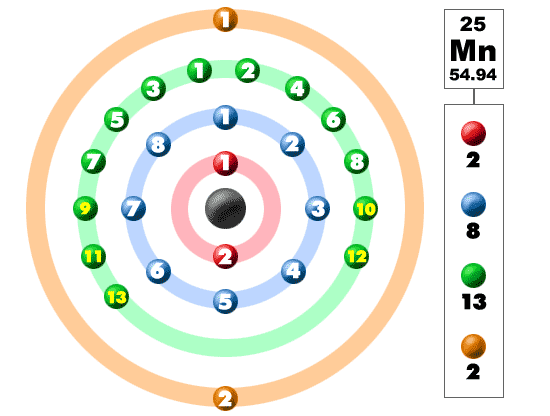
Manganese' symbol is Mn, with an atomic number of 25. The element Manganese has isotopes this meaning the amount of nuetrons change. It is made up of 25 protons and electrons and 30 Neutrons. The diagram to your left shows the make-up of these three things in a colour coded order.
This is the Complete Electron Configuration Notation for the Atom.
Properties
Physical properties are any matter which determine the matter and energy within their interactions. Manganese' five most common physical properties are:
Physical Property:1. Melting Point
2. Boiling Point 3. Standard State 4. Density 5. Heat of Fusion |
Example:1. 1246°C is Manganese' Melting Point
2. Manganese has a boiling point of up to 2061°C 3. Solid is Manganese' Standard State 4. Both density being 7.21g/cm^3 and liquid density being 5.95g/cm^35. Manganese has a physical property called Heat of fusion which is 12.91kJ/mol |
Chemical properties are a property used to characterize materials in reactions that change their identity. Manganese' three most common chemical properties are:
Chemical Property:1. Reaction with water
2. Oxygen reactions 3. Flame and Heat reaction |
Example:1. Manganese does not react with neutral water but does reaction with acidic water.In neutral water manganese will decompose and when added to acidic water it will dissolve and dilute.
2. Manganese when pure is reactive, but when made a powder will burn in oxygen.
3. When manganese is exposed to flames, manganese dust and chemicals become highly flammable.
|